Seek and ye shall find
The risk of over-diagnosis of disease when using periodontal diagnostic techniques for dental implants
As one of the largest specialist referral dental practices in the country and with more than 20 years of experience managing patients with dental implants, we have seen many surgical and restorative “trends” come and go (often on more than one occasion). These include: block bone grafting, guided surgery, distraction osteogenesis, cemented restorations, platform switching and composite resin bridgework.
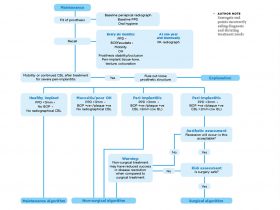
In the last few years, the most concerning of these, for us, has been the rise of peri-implantitis, both in terms of perceived incidence and proposed management. Well-meaning practitioners (generalist and specialist), together with representative bodies in the profession, have advocated using periodontal indices to produce algorithms for monitoring dental implants (See ADI algorithm overleaf).
The algorithms dictate when and how “ailing” implants should be treated. Such algorithms are only effective if periodontal indices are a reliable predictor of treatment needs. If not, there is a very real concern that many patients will, and are being, unnecessarily treated, often at great expense and with significant mental anguish.
Our prosthodontists, periodontists and oral surgeons have analysed and discussed at length the literature available on peri-implantitis.
This article and the one that follows are taken from a more extensive paper which investigates in detail the evidence to support the use of periodontal indices in implant monitoring. The paper has been submitted and accepted for publication in Periodontology 2000.
Clinical evidence does not support the published data
Osseointegrated dental implants have been used for more than 50 years and have truly revolutionised modern clinical dentistry. This treatment modality has given clinicians the possibility to give totally edentulous patients back their teeth and dignity.
In the same way, partial edentulism and single tooth gaps can be successfully managed with dental implants as a first choice of treatment to restore function and aesthetics without damaging remaining teeth. The use of dental implants is scientifically well documented and numerous long-term studies have shown predictable results with few serious complications. Moreover, due to better understanding of the biology of implants, improved components and clinical techniques, the literature has shown increasingly improved clinical outcomes with time (Figures 1, 2 and 3).
In spite of this, critical reports claim that up to 56 per cent of dental implants show signs of disease when examined with periodontal indices such as probing pocket depth (PPD), bleeding on probing (BoP) and radiographic marginal bone loss (MBL)1,2. Extrapolation of these findings based on the rationalities of periodontal diagnostics tools, suggests a future tsunami of “peri-implantitis”, i.e. severe marginal bone resorption, and subsequent implant losses (Figure 4).
Based on the same logic, it is recommended that the diseased implants need to be treated with non-surgical or surgical interventions to “resolve the disease”3. In an editorial in a highly ranked dental journal4, a restricted use of dental implants was advocated. This advice was based on the findings from a series of studies5–7 in the same journal claiming high levels of peri-implant disease among a large group of patients. If correct, our waiting rooms would be crowded by previously treated implant patients, now suffering from peri-implantitis.
In addition, the literature would be dominated by clinical reports with catastrophic failure rates. On the contrary, large numbers of suffering implant patients are missing and the scientific literature repeatedly demonstrates dental implants to be very successful with failure rates around 5 per cent after 10 years with about 2-3 per cent of implants affected by severe marginal bone loss and so called peri-implantitis8.
One may argue that most scientific follow-up studies are made in well-controlled academic environments and for that reason the results do not reflect clinical reality out in the field. However, it is notable that from the series of studies claiming high levels of peri-implant disease as referred to in the critical editorial, the same research group demonstrated only a
1.4 per cent early failure rate for 11,311 implants placed in 2,765 patients in the hands of more than 800 clinicians, and a 3 per cent total failure rate for a subgroup of 596 patients examined after nine years of function6. However, in spite of this, the authors of the study6 concluded that “implant failure is not an uncommon event”. In reality most of us would rejoice over such outstanding results.
Risks with surrogate endpoints in clinical research
How is it even possible to reach so different conclusions based on the same knowledge?
The answer is that the different research groups use different endpoints in their research9. Most clinical implant investigations are using true endpoints as primary parameters and measuring clinical outcomes in terms of definitive events, i.e. loss and removal of the implants and of the associated prosthetic restorations, whereas others are using surrogate endpoints (BoP, increased PPD) that, according to their hypothesis, will eventually lead to a definitive event (implant loss).
One example is one of the first studies reporting on alarming levels of peri-implant disease based on BoP and bone loss occurring after the first year of function10. According to the authors, about 56 per cent of all implants were affected by disease and had an expected poor prognosis. However, when analysing the same patient cohort nine years later, there were no differences between “affected” and healthy implants with regard to implant failures11. Moreover, 91.4 per cent of the implants in the patients diagnosed with peri-implantitis showed either no or minimal annual bone loss during the nine years from the diagnosis. Hence, the surrogate endpoints used by the authors, namely bone loss associated with bleeding on probing, were shown to be poor predictors of future bone loss and implant failure and consequently poor indicators of treatment needs (Table 1). With such a periodontal approach for the diagnosis of peri-implant diseases, there is an obvious risk that patients will be subjected to unnecessary treatment, which is unacceptable from an ethical point of view as it may create iatrogenic as well as economic damage to the patients.
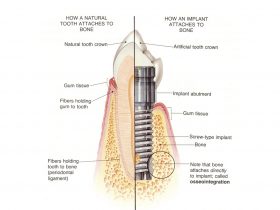
Teeth versus implants (Figure 5)
The periodontal complex is the result of millions of years of evolution and is built up by highly differentiated and specialised tissues. Osseointegration on the other hand, is the result of a foreign body reaction to an implant. The soft and bone tissue interfaces consist of lowly differentiated scar tissues12,13. Hence, from a morphological point of view, the tooth and the implant represent two different entities, which cannot be directly compared.
Inflammation and marginal bone loss at teeth, i.e. gingivitis and periodontitis, are considered as infectious and biofilm-mediated conditions. These are diagnosed by probing, where bleeding (BoP) indicates gingivitis and increased pocket depths (PPD) with bleeding (BoP) and bone loss indicate periodontitis. Removal of the biofilm and bacteria formation will resolve the inflammation/infection. This means that clean teeth will not show any signs of bleeding and increased pocket depths.
Dental implants and mucosa-piercing components are placed in edentulous areas of the jaws during one or two surgical interventions. It is well documented that some marginal remodelling will occur during the first year as a response to surgery, piercing of the mucosa and loading. For a large group of implants, the average bone loss during the first year in function varies from 0.5 to 1.5 mm, mainly depending on the geometry of the implant 14,15. After the first year in service, small changes of marginal bone loss are observed over the following years of follow-up.
However, if making a frequency distribution of bone loss, some implants will show more bone loss than other implants 16. The reasons for bone loss can be physiological atrophy after tooth extraction, non-optimal surgery and prosthetic design, overload, thin bone, soft bone, cement residues just to mention a few (Figure 6). This means that healthy and well-functioning dental implants, in contrast to teeth, can show non-infectious marginal bone resorption. Even in a hypothetical environment without bacteria, there will be bone loss around implants. For this reason, a zero-tolerance approach to bleeding, pocket depths and some marginal bone loss does not appear to be justified.
It is often pointed out that patients with a history of severe periodontitis are more susceptible to marginal bone loss at dental implant (Figure 7). Some authors see this as a proof of a bacterial and biofilm-induced process. However, another plausible explanation is that this is related to the immunologic foreign-body response to plaque and implant components in these patients, which for some reason is exaggerated 11.
The use of periodontal diagnostic techniques at implants
In analogy with teeth, the rationale for using a dental probe round implants is to identify “affected” implants based on the presence of any BoP and PPDs deeper than 4 mm, which in combination with any radiographic bone loss, would indicate disease. In reviewing the literature, it is evident that BoP may be detected in more than 90 per cent of well-functioning dental implants 17. Likewise, PPDs over 4 mm are frequent round healthy implants (Figure 8). The depth of the peri-implant pocket is not immediately pathological; it is, of course, dependant on the thickness of the mucosa and how deep the implant was placed.
As discussed earlier, literally all implants will show some bone loss over time as a result of many other factors than simply the “catch all” of biofilm-mediated resorption. A recent review of the literature, where studies on the use of periodontal probes to diagnose peri-implant health have been used, 17 concluded that:
- Probing pocket depth values of >4 mm at dental implants cannot be seen as a sign of pathology or an alarming signal regarding the conditions of the peri-implant tissues.
- An increase of probing pocket depth values over time is not necessarily associated with loss of supporting bone around dental implants. Therefore, probing does not appear to be a reliable tool for the assessment of peri-implant marginal soft- and hard-tissue conditions.
- From a biomaterials science point of view, osseointegration is a foreign-body reaction. As a consequence, bleeding on probing does not always indicate the presence of acute inflammation in the peri-implant mucosa, but may rather reflect the nature of the scar tissue–implant interface. Therefore, bleeding on probing does not appear to be a predictor for future loss of tissue support. Similarly, absence of bleeding on probing does not appear to be a predictor of future stability. Hence, probing pocket depth and bleeding on probing cannot be considered to be reliable tools for monitoring peri-implant health and disease.
- Radiographic evaluation of crestal bone levels over time seems to be the most reliable tool to identify those implants undergoing continuous bone loss and therefore in need of treatment.
- A single episode of bone loss does not necessarily call for treatment unless associated with clear signs of inflammation, such as profuse bleeding/suppuration and discomfort at pressure/palpation.
Conclusion
This first of a two-part paper has been written to address the possibility that using periodontal indices to monitor dental implant health may not only be unnecessary but in many circumstances may result in patients receiving treatment that they don’t need.
In part two, we will look at proposed guidelines for routine follow up of dental implants and how true peri-implantitis lesions may be managed.
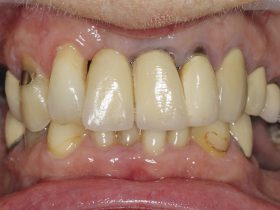
- FIGURE 1: Failing crown and bridgework
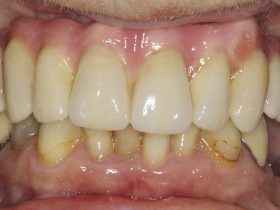
- FIGURE 2: Fixed full arch bridge porcelain finish
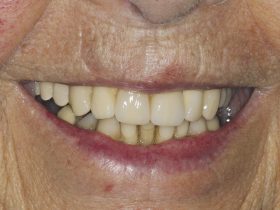
- FIGURE 3: Appearance, function and dignity predictably returned
- Article from the Telegraph website (14 July 2014)
- Figure 5: A comparison of how a natural tooth and an implant attaches to the bone
- FIGURE 6: Bone remodelling is to be expected round healthy implants and is not necessarily due to biofilm mediated pathology
- FIGURE 7: Are periodontitis patients more susceptible to periimplantitis
- FIGURE 8: Perfectly healthy implant with more than 4mm pocket depth and BoP
- ADI maintenance algorithm
REFERENCES
- Lindhe J, Meyle J. Peri-implant diseases: consensus report of the sixth European workshop on periodontology. J ClinPeriodontol 2008: 35: 282–285.
- Lang NP, Berglundh T. Peri-implant diseases: where are we now? Consensus of the Seventh European Workshop on periodontology. J ClinPeriodontol 2011: 38: 178–181.
- Ucer C, Wright S, Scher E, West N, Retzepi M, Simpson S, Slade K, Donos N. ADI guidelines. On peri-implant itoring and maintenance. 2013: http://www.adi.org.uk/resources/guidelines_and_papers/peri-implant/
- Giannobile WV, Lang NP. Are dental implants a panacea or should we betterstrive to save teeth? J Dent Res 2016: 95:5–6.
- Derks J, Håkansson J, WennströmJL, Klinge B, Berglundh T. Patient-reported outcomes of dental implant therapy in a large randomly selected sample. Clin Oral Implants Res 2015: 26: 586–591.
- Derks J, Håkansson J, WennströmJL, Tomasi C, Larsson M,Berglundh T. Effectiveness of implant therapy analyzed in a Swedish population: early and late implant loss. J Dent Res2015: 94(3 Suppl.): 44S–51S.
- Derks J, Schaller D, Håkansson J, WennströmJL, Tomasi C,Berglundh T. Effectiveness of implant therapy analyzed in a Swedish population:
prevalenceof peri-implantitis. J DentRes 2016: 95: 43–49. - Albrektsson T, Buser D, Sennerby L. Crestal bone loss and oral implants. ClinImplant Dent Relat Res 2012: 14: 783–791.
- Psaty BM, Weiss NS, Furberg CD, et al. (1999). “Surrogate end points, health outcomes, and the drug approval process for the treatment of risk factors for cardiovascular disease”.JAMA. 282 (8): 786–790.
- Fransson C, Lekholm U, Jemt T, Berglundh T. Prevalence of subjects with progressive bone loss at implants. Clin OralImplants Res 2005: 16: 440–446.
- Jemt T, Sundén Pikner S, K Gröndahl. Changes of marginal bone level in patients with ‘progressive bone loss’ at Brånemark System implants: a radiographic follow-up study over an average of 9 years. ClinImplant Dent Relat Res2015: 17: 619–628.
- Donath K. Pathogenesis of bony pocket formation around dental implants. J Dent Assoc S Afr 1992; 47:204–208.
- Albrektsson T, Dahlin C, Jemt T, Sennerby L, Turri A, WennerbergA. Is marginal bone loss around oral implants the result of a provoked foreign body reaction? ClinImplantDent Relat Res 2014: 16: 155–165.
- Oh TJ, Yoon J, Misch CE, Wang HL. The causes of early implant bone loss: myth or science? J Periodontol 2002: 73:322–333.
- Laurell L, Lundgren D. Marginal bone level changes at dental implants after 5 years in function: a meta-analysis. ClinImplant Dent Relat Res 2011: 13: 19–28.
- Pikner SS, Gröndahl K, Jemt T, Friberg B. Marginal bone loss at implants: a retrospective, long-term follow-up of turned Brånemark System implants. ClinImplant Dent Relat Res. 2009 Mar;11(1):11-23.
- Coli P, Christiaensen V, Sennerby L, De Bruyn H. Reliability of periodontal diagnostic tools for monitoring peri-implant health and disease. Periodontology 2000, in press
About the authors
- Dr Pierluigi Coli is a specialist in periodontics and prosthodontics at Edinburgh Dental Specialists.
- Dr Kevin Lochhead is a specialist in prosthodontics and principal dentist at Edinburgh Dental Specialists.
- Professor Lars Sennerby is a visiting professor in implantology at Edinburgh Dental Specialists.
Verifiable CPD Questions
AIMS and Objectives:
- To draw attention to the questionable use of periodontal indices to monitor dental implants
- To explained why periodontal probing may not be justified
- To explain why there is contention within the profession regarding monitoring of implants
- To draw attention to the potential for overdiagnosis and overtreatment.
LEARNING OUTCOMES:
- To understand that most implants will bleed on probing
- To understand that pocket depth is not an accurate measure of treatment needs
- To understand that bacterial biofilm induced bone loss is only one of many causes of bone loss round implants.




Comments are closed here.